A Look at Pennsylvania Clinical Trials in April 2026
April turned out to be a relatively quiet month for new clinical research openings across Pennsylvania. Only 15 new studies were launched statewide, down from 51 in March and 55 in February. The smaller count is not a pattern that reflects anything concerning in the state's research ecosystem; some months simply run lighter than others. It does make this a useful month for looking at what opened and who is running the studies, without drowning in volume.
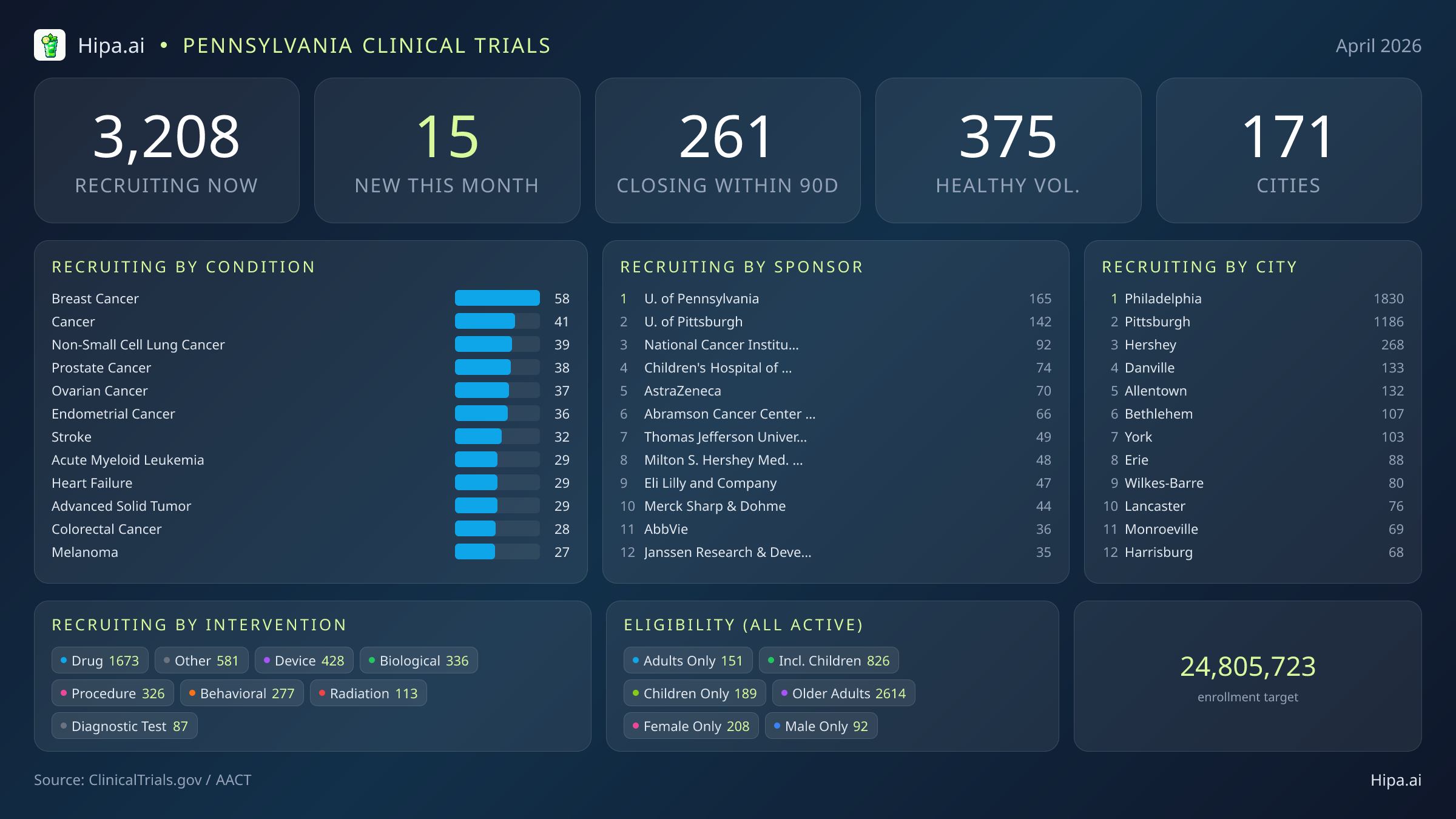
Scale across the state remains substantial regardless of the monthly rate of new launches. Pennsylvania has 3,208 currently recruiting trials across 171 cities and 1,732 research sites, with 261 of those scheduled to close enrollment within the next 90 days. The closing-soon count is often the more time-pressured one for patients. Openings come on a rolling basis, but enrollment windows that shut in 90 days are finite, and once they close, you are waiting for the next cycle.
For anyone in Pennsylvania considering participation, the directory of paid clinical trials in Pennsylvania shows current listings filtered by city, condition, age, and healthy volunteer status. Most active research clusters in Philadelphia (2,072 recruiting) and Pittsburgh (1,325 recruiting), but the state has 171 cities running research sites, with smaller concentrations in Hershey (289), Danville (141), Allentown (134), Bethlehem (108), and plenty of others.
Notable New Launches
Of the 15 April openings, a handful stand out for either enrollment scale or sponsor prominence.
Novartis Pharmaceuticals added a Yardley site to NCT07517263, the open-label long-term extension study for pelacarsen (TQJ230), which targets lipoprotein(a)-driven cardiovascular disease. This extension phase enrolls 5,700 participants who previously completed the preceding Phase 3 double-blind protocol (CTQJ230A12301).
Incyte Corporation opened NCT07522073 in Philadelphia, a Phase 3 trial evaluating chemotherapy with or without INCB161734 in patients with previously untreated, KRAS G12D-mutated metastatic pancreatic ductal adenocarcinoma. Target enrollment is 588.
Kinaset Therapeutics launched NCT07532265 in Pittsburgh, a Phase 2 dose-ranging study of frevecitinib (KN-002) in patients with severe asthma that is not well-controlled on standard treatment. The 12-week protocol targets 512 participants.
The VA Office of Research and Development is running NCT07507747 in Philadelphia, which tests an adaptive, peer-supported mobile health intervention for primary-care veterans experiencing psychological distress, including depression, PTSD, anxiety, and sleep problems. Target enrollment is 384.
Iantrek opened NCT07520175 in Cranberry Township, which follows patients after bio-reinforced canaloplasty for open-angle glaucoma. The enrollment target is 300, and the study assesses both the procedure's effect on intraocular pressure and how long that effect lasts.
Sponsor Mix and Research Focus
Three of the 15 new April trials are sponsored by the University of Pennsylvania and two by the University of Pittsburgh. Industry sponsors (Novartis, Incyte, Kinaset Therapeutics, and Iantrek) account for four more. The remaining six come from other sources, including the VA and smaller investigator-led studies.
By intervention type, the 15 were split across five device trials, five drug trials, three behavioral interventions, one procedure, and two classified as other. Condition coverage is broad: psychological distress (two studies), cancer, cardiovascular disease (including one specifically on cardiovascular disease with elevated lipoprotein(a)), cerebral palsy, chronic kidney disease, coronary artery disease, and several coronary calcification-related conditions.
City distribution is concentrated. Philadelphia and Pittsburgh each hosted six of the 15 new trials. Cranberry Township, Kingston, Yardley, and York got one each. That distribution tracks with where Pennsylvania's academic medical centers sit, and the smaller-city placements are typically specialty work (surgical, cardiovascular, or device studies running out of regional centers).
Who Can Participate
For patients without a specific diagnosis who still want to contribute to research, there are meaningful options. Of the 15 new April trials, two are recruiting healthy volunteers. Across all currently recruiting trials statewide, 375 are open to healthy participants.
Age eligibility in the new April studies skews toward adults but is not exclusively adult. Three of the new trials include children. Thirteen include older adults, which tracks with the condition mix in the month's new openings.
On the closing-soon side, 48 of the 261 trials winding down in the next 90 days are recruiting healthy volunteers. The most common conditions among those closing are cancer, opioid use disorder, chronic pain, breast cancer, sleep, type 2 diabetes, schizophrenia, gastric cancer, stroke, and colorectal cancer.
Looking Ahead
April is one data point in Pennsylvania's broader research calendar. What matters for patients tracking participation is that the overall scale of active trials keeps growing and that specific-condition trials move through the pipeline at different rates. Hipa.ai and ClinicalTrials.gov both track the current state in near real-time, which is useful whether you are weighing participation personally or just following what research is active in your area.
